Medical electronics—devices that use electronic systems for diagnosis, monitoring, and therapy—have become the backbone of modern healthcare. From wearable ECG monitors to AI-enabled imaging systems, this sector is expanding rapidly, driven by aging populations, digital health adoption, and regulatory innovation.
This data-driven guide breaks down the market size, growth rates, cost metrics, and performance benchmarks shaping medical electronics in 2024–2026.
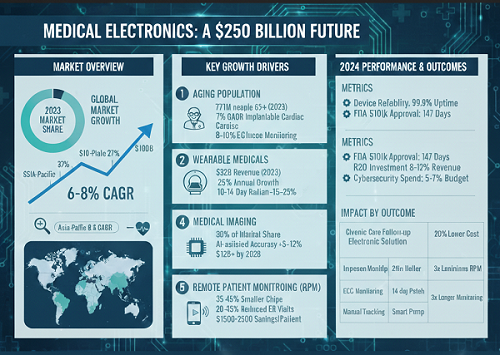
Global Market Size: $160+ Billion Industry Growing at 6–8% CAGR
According to recent industry analysis by Dataintelo, the global medical electronics market reached approximately $168 billion in 2023 and is estimated to hit nearly $180 billion in 2024. The market is projected to grow at a 6.5%–7.8% compound annual growth rate (CAGR) through 2030, potentially surpassing $250 billion by the end of the decade.
Regional Breakdown (2023 Estimates)
| Region | Market Size (USD) | Market Share | Growth Rate (2024–2028) |
| North America | $62B | 37% | 6.2% CAGR |
| Europe | $45B | 27% | 5.8% CAGR |
| Asia-Pacific | $44B | 26% | 8.5% CAGR |
| Rest of World | $17B | 10% | 6.0% CAGR |
Asia-Pacific’s 8.5% CAGR is the highest globally, driven by China and India’s healthcare digitization investments, which increased by 12–15% annually since 2021.
5 Data-Backed Growth Drivers in Medical Electronics
1. Aging Population: 771 Million People Aged 65+
In 2023, the global population aged 65+ reached 771 million, representing 9.7% of the total population. By 2030, this will exceed 1 billion people, increasing demand for:
- Implantable cardiac devices (projected 7% CAGR)
- Glucose monitoring systems (8–10% CAGR)
- Portable diagnostic electronics (6%+ CAGR)
Older adults account for approximately 60% of medical device usage globally.
2. Wearable Medical Electronics: 25% Annual Growth
Medical-grade wearables generated approximately $32 billion in revenue in 2023, up from $25 billion in 2021—a 28% increase in two years.
Key performance benchmarks:
- Continuous glucose monitors (CGMs) improved accuracy to MARD ≤8% in 2023, compared to 10–12% in 2018.
- Battery life in wearable ECG patches increased from 3–5 days (2019) to 10–14 days (2024).
- Remote patient monitoring (RPM) reduced hospital readmissions by 18–25% in chronic care programs.
3. Medical Imaging Electronics: High-Cost, High-Precision Systems
Medical imaging electronics (MRI, CT, ultrasound) represent roughly 30% of total medical electronics revenue, or about $50–55 billion annually.
Cost benchmarks:
- MRI system: $1–3 million per unit
- CT scanner: $500,000–$2 million
- Portable ultrasound: $5,000–$50,000
Performance improvements (2020–2024):
- AI-assisted radiology improved diagnostic accuracy by 5–12%
- Image processing speeds increased by 30–40%
- Radiation exposure in modern CT systems reduced by 15–25%
4. Semiconductor Integration: 40% Smaller, 30% More Efficient
Medical electronics depend heavily on specialized semiconductors.
Between 2018 and 2024:
- Chip size in implantable devices decreased by 35–45%
- Power consumption reduced by 25–30%
- Device battery life extended by 20–50%, depending on application
The medical semiconductor segment alone surpassed $8 billion in 2023, with projections exceeding $12 billion by 2028.
5. Remote Patient Monitoring (RPM): Cost Reduction of 20%+
Hospitals using electronic RPM systems reported:
- 20–25% reduction in emergency visits
- 15–20% shorter hospital stays
- Average savings of $1,500–$2,500 per patient annually in chronic disease management
In 2023, over 30 million patients worldwide were enrolled in RPM programs—up from 18 million in 2020, representing a 67% increase in three years.
2024 Performance Metrics: Reliability, Compliance & Investment
Device Reliability
- Medical electronics must meet 99.9%+ operational uptime
- Implantable devices often target failure rates below 0.1% annually
- Mean Time Between Failures (MTBF) for hospital-grade equipment: 50,000–100,000 hours
Regulatory Timelines
- Average FDA 510(k) clearance time (2023): 147 days
- PMA approval process: 180–320 days
- R&D spending typically equals 8–12% of annual revenue for leading manufacturers — a reflection of how complex and resource-intensive medical device product development has become.
Cybersecurity Investment
In 2024:
- Healthcare cyberattacks increased by 13% year-over-year
- 60% of medical device recalls involved software vulnerabilities
- Companies now allocate 5–7% of product development budgets to cybersecurity compliance
Cost vs. Outcome: Quantifying ROI in Medical Electronics
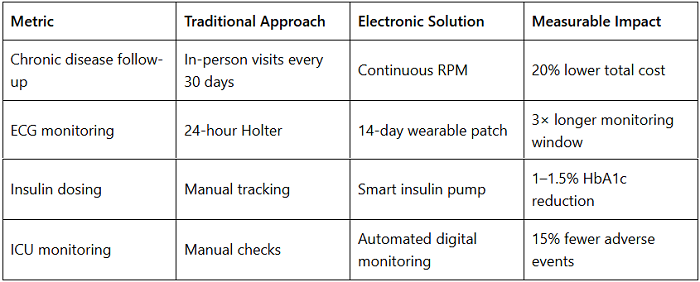
Hospitals adopting integrated electronic monitoring systems reported ROI within 12–24 months, depending on scale.
Key Statistics at a Glance (Medical Electronics Industry)
| Metric | Value | Timeframe / Context |
| Global Market Size | $180 Billion | 2024 estimate |
| Projected CAGR | 6.5%–7.8% | 2024–2030 forecast |
| Imaging Electronics Market Share | 30% of total market | 2023–2024 |
| Wearable Segment Revenue | $32 Billion | 2023 |
| Wearable Growth Rate | 28% increase | 2021–2023 |
| RPM Adoption Growth | 67% increase | Since 2020 |
| Annual Savings per RPM Patient | $1,500–$2,500 | Chronic care programs |
| Semiconductor Chip Size Reduction | 35–45% smaller | 2018–2024 |
| Reduction in Hospital Readmissions | 20–25% decrease | With monitoring electronics |
| Clinical System Uptime Requirement | 99.9% minimum | Industry benchmark |
| Average FDA 510(k) Review Time | 147 days | 2023 average |
| R&D Investment | 8–12% of annual revenue | Leading manufacturers |
| Cybersecurity Budget Allocation | 5–7% of development budget | 2023–2024 |
Forecast: What to Expect by 2030
By 2030:
- Market size projected to surpass $250 billion
- AI-integrated devices expected in 70% of imaging systems
- Remote monitoring adoption could exceed 60 million patients globally
- Wearables may represent 25% of total medical electronics revenue, up from ~18% in 2023
- Device miniaturization could reduce implant size another 20–30%
Healthcare providers increasingly measure value in cost per patient outcome, not just equipment price. Electronics that deliver measurable reductions in hospital stays (15–20%), readmissions (20%+), and adverse events (10–15%) will dominate procurement decisions.
Conclusion: A $250 Billion Opportunity Driven by Measurable Outcomes
Medical electronics is no longer a niche engineering field—it is a $180+ billion global industry growing at nearly 7% annually. With 30% of revenue from imaging, $32 billion from wearables, and 67% growth in remote monitoring since 2020, the sector’s expansion is strongly supported by data.
Quantified outcomes—20% lower healthcare costs, 15% shorter hospital stays, 35% smaller chips, and 99.9% system reliability standards—demonstrate that innovation in medical electronics directly translates into measurable clinical and financial impact.
By 2030, as the market approaches $250 billion, the winners will be companies that deliver not just advanced electronics—but verifiable, statistically proven performance improvements across cost, accuracy, safety, and patient outcomes.
For more information, visit: https://dataintelo.com/report/medical-electronics-market